Product Name: GP130, Protein Dimer
Predicted Molecular Weight: 156 kDa
SDS PAGE Molecular Weight: The migration range of the dimer protein with glycosylation under non-reducing conditions is >190 kDa on SDS PAGE.
Protein Construct: Gp130 dimer protein contains a gp130 extracellular domainfused with a proprietary dimer motif followed by a tandem His-Avi tag at the C-terminus.
Human glycoprotein 130 (gp130), is a transmembrane protein and a member of the class of tall cytokine receptors. Gp130 is also known as Interleukin 6 Cytokine Family Signal Transducer (IL6ST), Cluster of Differentiation 130 (CD130), CDW130, and Interleukin-6 Receptor Subunit Beta (IL6Rb). Gp130 serves as a shared signal transducing subunit of the receptor complexes for at least nine human cytokines including: interleukin-6 (IL-6), interleukin-11 (IL-11), interleukin-27 (IL-27), leukemia inhibitory factor (LIF), ciliary neurotrophic factor (CNTF), oncostatin M (OSM), cardiotrophin-1 (CT-1), cardiotrophin-like cytokine (CLC/CLCF-1), and neuropoietin (NP) that mediate highly diverse biological processes. Gp130 can form homodimers and heterodimers with other cytokine receptors (i.e., IL-6 receptor alpha (IL-6Ra)) in response to cytokine binding. The homodimerization or heterodimerization of gp130 is key to initiating intracellular signaling pathways, resulting in the activation of gp130-associated JAKs (JAK1, JAK2, and TYK2). The extracellular domain of gp130 includes an N-terminal immunoglobulin-like (Ig-like) domain (D1), a cytokine-binding homology region (CHR, D2D3), and three membrane-proximal fibronectin type III domains (FNIII, D4 to D6) followed by a transmembrane domain and cytoplasmic domain. It has been found that dysregulation of gp130 expression and signaling mediates progression for multiple types of cancer and autoimmune diseases. Inhibition of gp130 activity offers a potential and promising approach to cancer and autoimmune disease therapy.
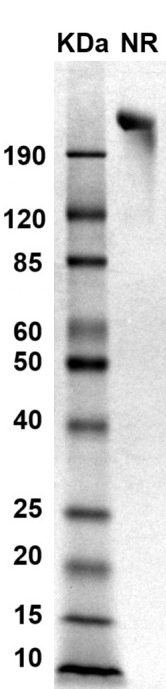 MW: Molecular Weight marker reduced condition
NR: gp130 dimer under non-reducing condition
MW: Molecular Weight marker reduced condition
NR: gp130 dimer under non-reducing condition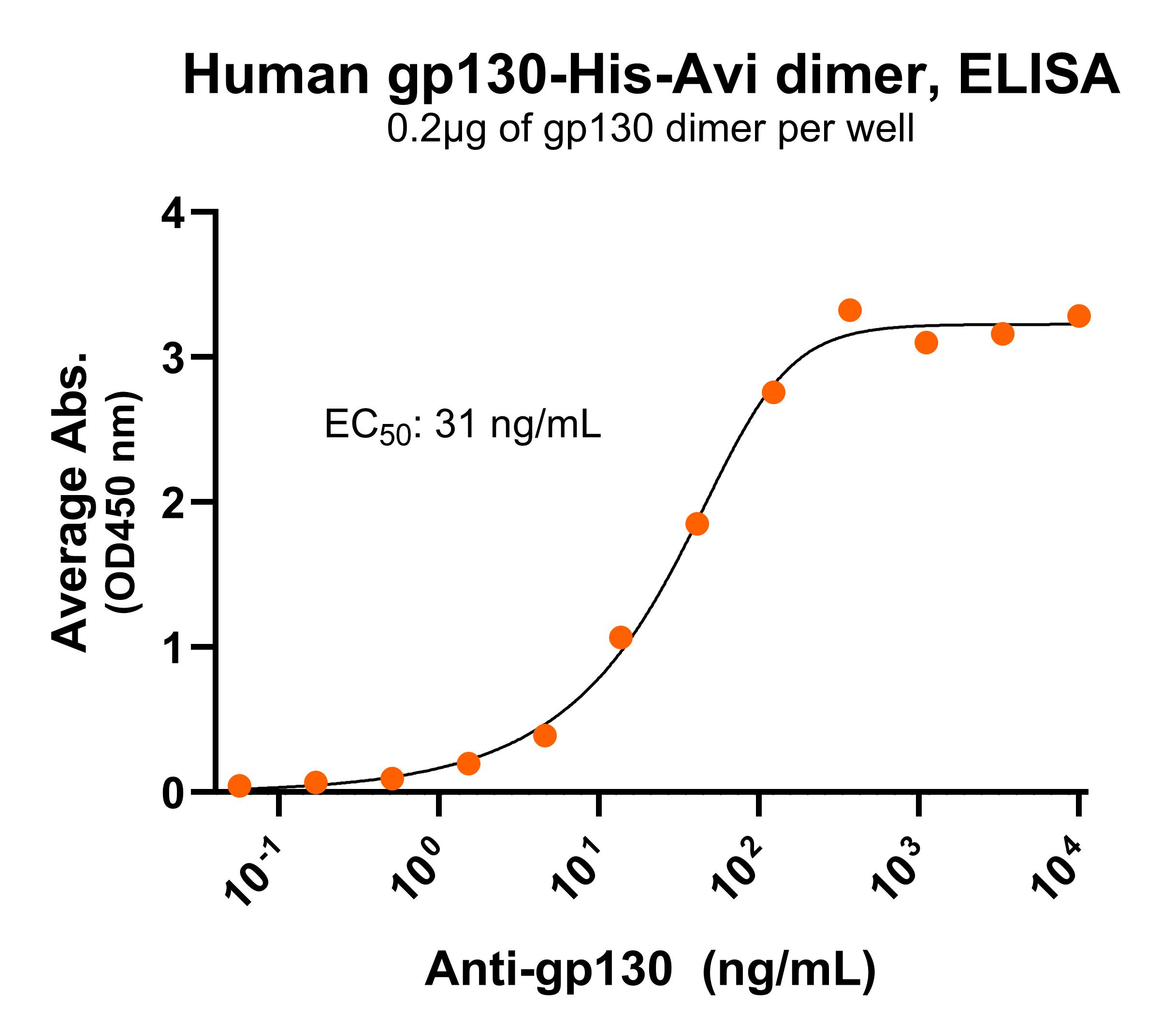 Immobilized human gp130dimer protein, His-Avi Tag (Cat. No. CSP-24081-03) at 2 μg/mL (100 μL/well) can bind anti-human gp130 monoclonal antibody, with half maximal effective concentration (EC50) range of 15.6-62.4 ng/mL (QC tested).
Immobilized human gp130dimer protein, His-Avi Tag (Cat. No. CSP-24081-03) at 2 μg/mL (100 μL/well) can bind anti-human gp130 monoclonal antibody, with half maximal effective concentration (EC50) range of 15.6-62.4 ng/mL (QC tested).