Product Name: PD-L1, Protein Dimer
Predicted Molecular Weight: 102 kDa
SDS PAGE Molecular Weight: The migration range of the dimer protein with glycosylation under non-reducing condition is ~190 kDa on SDS PAGE.
Protein Construct: PD-L1 protein dimer contains a PD-L1 extracellular domainfused with a proprietary cis-dimer motif followed by a Fc tag at the C-terminus.
Programmed death-ligand 1 (PD-L1), is a Type I transmembrane protein in the immunoglobulin superfamily and a member of the B7 Family of ligands. PD-L1 is also known as cluster of differentiation 274 (CD274), B7 homolog 1 (B7H1, B7-H1), PDCD1L1, PDCD1LG1, and CD274 molecule. PD-L1 contains an extracellular domain with a distal immunoglobulin V-like (Ig-V-like) domain and proximal immunoglobulin C-like (Ig-C-like) domain, a transmembrane domain, and a cytoplasmic domain. PD-L1 is expressed on T cells, NK cells, macrophages, myeloid DCs, B cells, epithelial cells, and vascular endothelial cells. PD-L1 serves as an immunosuppressive ligand for PD-1 and the overexpression of PD-L1 on many tumor cells can prevent the immune system from attacking tumors. Inhibition of the interaction between PD-1 and PD-L1 can enhance antitumor activity, which has led to a new class of drugs called PD-1 inhibitors to activate the immune system and treat certain types of cancer. PD-L1 is highly expressed in a variety of malignancies, particularly lung cancer. PD-L1 exists as both a monomer and a dimer. Therefore, a recombinant protein mimicking the PD-L1 dimer conformation can be crucial for cancer therapeutic discovery. Mouse PD-L1, the murine homolog of human Programmed Death-Ligand 1 (PD-L1), is a critical immune checkpoint protein involved in regulating immune responses and is indispensable for studying immune checkpoint biology and immunotherapy. It is a species-specific tool essential for basic research, translational research and preclinical studies.
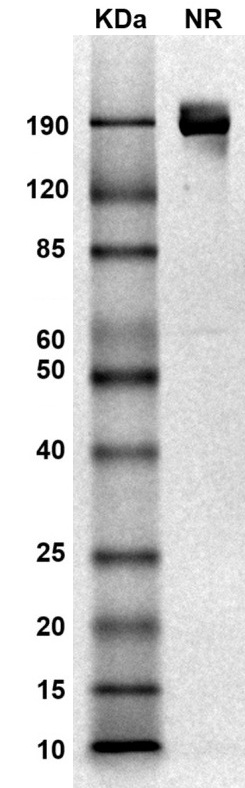 MW: Molecular Weight marker reduced condition
NR: PD-L1 dimer under non-reduced condition
MW: Molecular Weight marker reduced condition
NR: PD-L1 dimer under non-reduced condition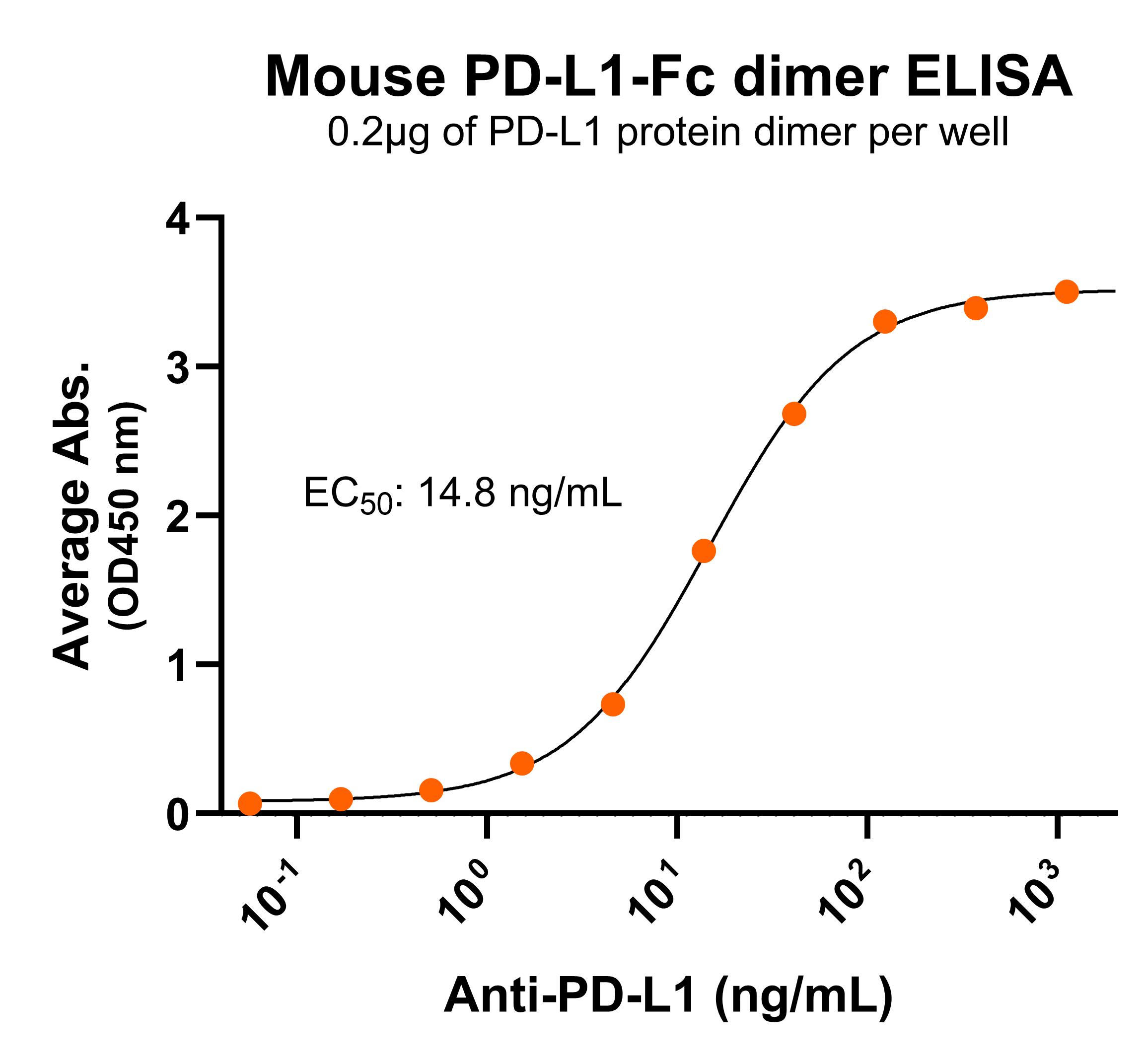 Immobilized mouse PD-L1-Fc protein dimer (CSP-25190-04) at 2 μg/mL (100 μL/well) can bind anti-mouse PD-L1 monoclonal antibody with half maximal effective concentration (EC50) range of 7.4-29.5 ng/mL (QC tested).
Immobilized mouse PD-L1-Fc protein dimer (CSP-25190-04) at 2 μg/mL (100 μL/well) can bind anti-mouse PD-L1 monoclonal antibody with half maximal effective concentration (EC50) range of 7.4-29.5 ng/mL (QC tested).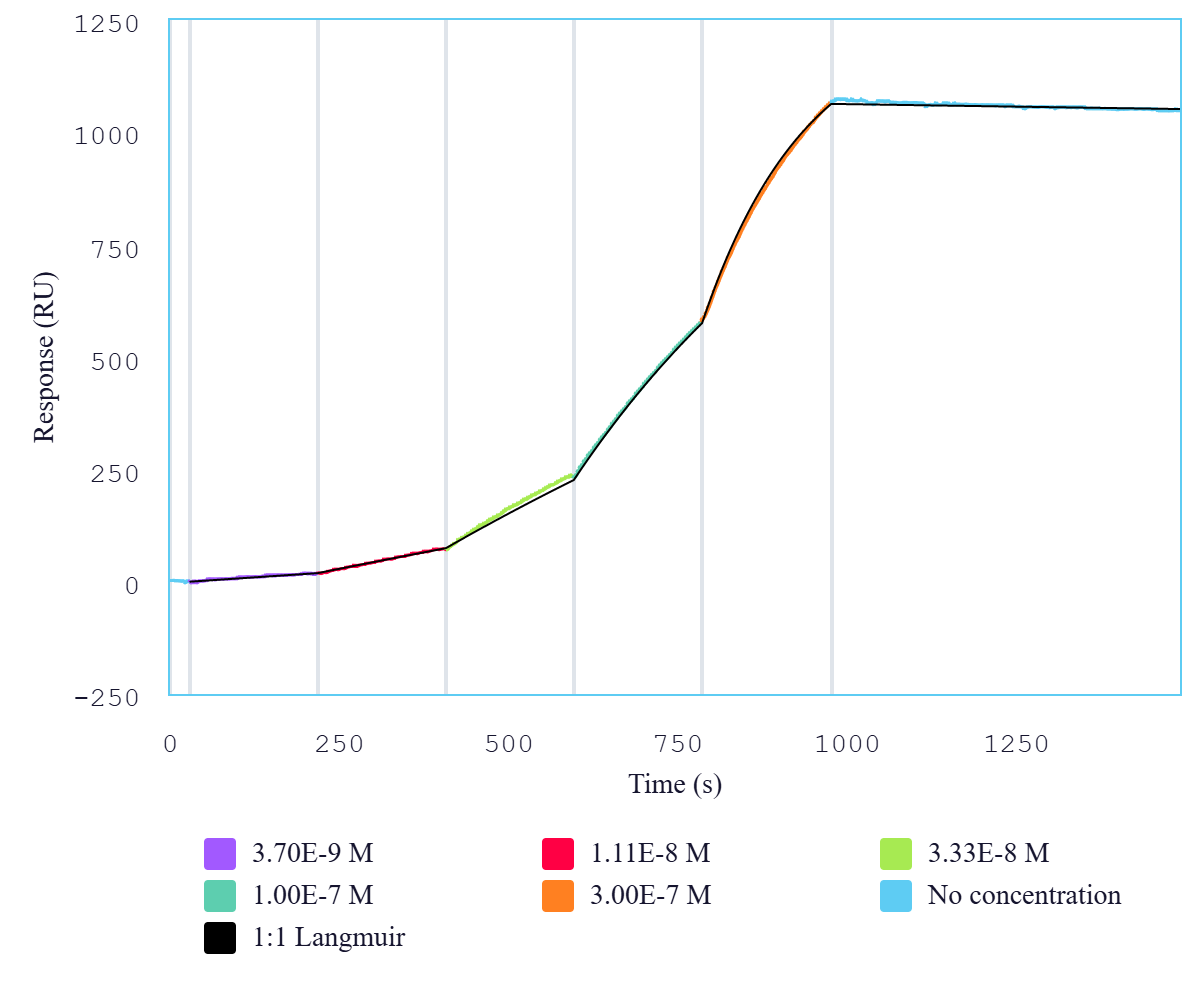 Immobilized mouse PD-L1 dimer protein, Fc tag (CSP-225190-04) can bind anti-mouse PD-L1 monoclonal antibody with a KD of 0.5-1.9 nM as determined by SPR.
Immobilized mouse PD-L1 dimer protein, Fc tag (CSP-225190-04) can bind anti-mouse PD-L1 monoclonal antibody with a KD of 0.5-1.9 nM as determined by SPR.